|
However, we do know that UV-A radiation is quite prevalent in natural daylight, and as a result, there are far more studies and population level studies that provide us with some level of understanding of the potential risks and harm that UV-A can cause. Because it is invisible, UV radiation can be especially dangerous as it does not induce a natural reaction to squint or look away the way we do with visible light. 
Significant precautions must be taken with all forms of ultraviolet energy, as all wavelengths of UV, including UV-A, UV-B and UV-C are presumed carcinogens, according to the US HHS. UV-C, on the other hand, is absorbed by the earth's ozone layer, and does not reach the ground. While solar radiation includes all wavelengths of UV energy, only UV-A and some UV-B pass through the earth's atmosphere. UV-A is Present in Daylight, UV-C is NotĪ common misconception is that the natural daylight includes all kinds of UV energy. Therefore, all evidence to date indicates that UV-A is not an effective method of disinfection as it is not able to target the DNA structures of pathogens.įor more information, see our page dedicated to UV-C LED technology. Below is a chart which shows that thymine and uracil do not absorb UV radiation at wavelengths higher than 300 nanometers.Īs the chart shows, UV-A does not have the ability to initiate dimerization in the way that UV-C radiation does. UV-C is unique in its ability to perform germicidal functions because of the wavelength susceptibility of thymine (uracil in RNA). Due to this alteration, when the pathogen attempts to replicate or reproduce, the genomic corruption prevents it from doing so successfully. When pathogens are exposed to UV-C wavelength radiation, double bonds that tie together thymine and adenine are destroyed in a process called dimerization, altering the structure of the pathogen's genome. UV-C is an effective germicidal wavelength because DNA and RNA are susceptible to damage at and around 265 nanometers. UV-C wavelengths have been the center of focus as an effective way to inactivate pathogens including viruses, bacteria, molds and fungi. Unlike UV-A, UV-C wavelengths occupy a much lower wavelength range of 100 nm to 280 nm. UV-C: Germicidal and Disinfection Applications In addition to intensity, the total exposure time is also a factor in UV-A curing applications. UV-A wavelengths are used for emulsion paint curing in screen printing, as well as epoxies for industrial applications as well as nail gels. As with fluorescence, 365 nm is a commonly used wavelength for curing. 
Generally, curing requires a significantly higher level of UV intensity, but is nonetheless accomplished using the same UV-A wavelengths. Unlike fluorescence, UV-A can also initiate chemical and structural changes in various materials, and is used in curing applications. For more information, see our article comparing 365 nm and 395 nm. Generally, both 365 and 395 nm will create fluorescence effects, but 365 nm will provide a "cleaner" UV effect with less visible light output, while 395 nm will have a small visible violet / purple component. The most common wavelengths for UV-A fluorescence are 365 nm and 395 nm. Therefore, it is even more important that you know what wavelength light source you will need for your particular application, and at the very least, to be aware of the differences between UV-A and UV-C radiation. UV-B wavelengths fall in between at 280 and 315 nanometers.īoth UV-A and UV-C are not visible to the naked eye, so it can be a bit counter-intuitive as you cannot visually see the difference between these two types of UV, the same way we can visually check if a light source is red or blue. 
UV-A encompasses wavelengths between 315 and 400 nanometers, while UV-C includes wavelengths from 100 and 280 nanometers. The wavelength value is measured in nanometers (nm) and is what determines the type of ultraviolet energy. Ultraviolet energy should first and foremost be identified by its wavelength. In this article, we discuss the primary differences between UV-A and UV-C radiation in terms of their applications and uses. It is very important, however, to distinguish between various types of ultraviolet energy, as each type has very different properties. Yet, when we think about ultraviolet, we tend to forget this, and simply categorize it as a range of wavelengths associated with its usefulness in fluorescence, curing, disinfection but also its potential carcinogenic effects. 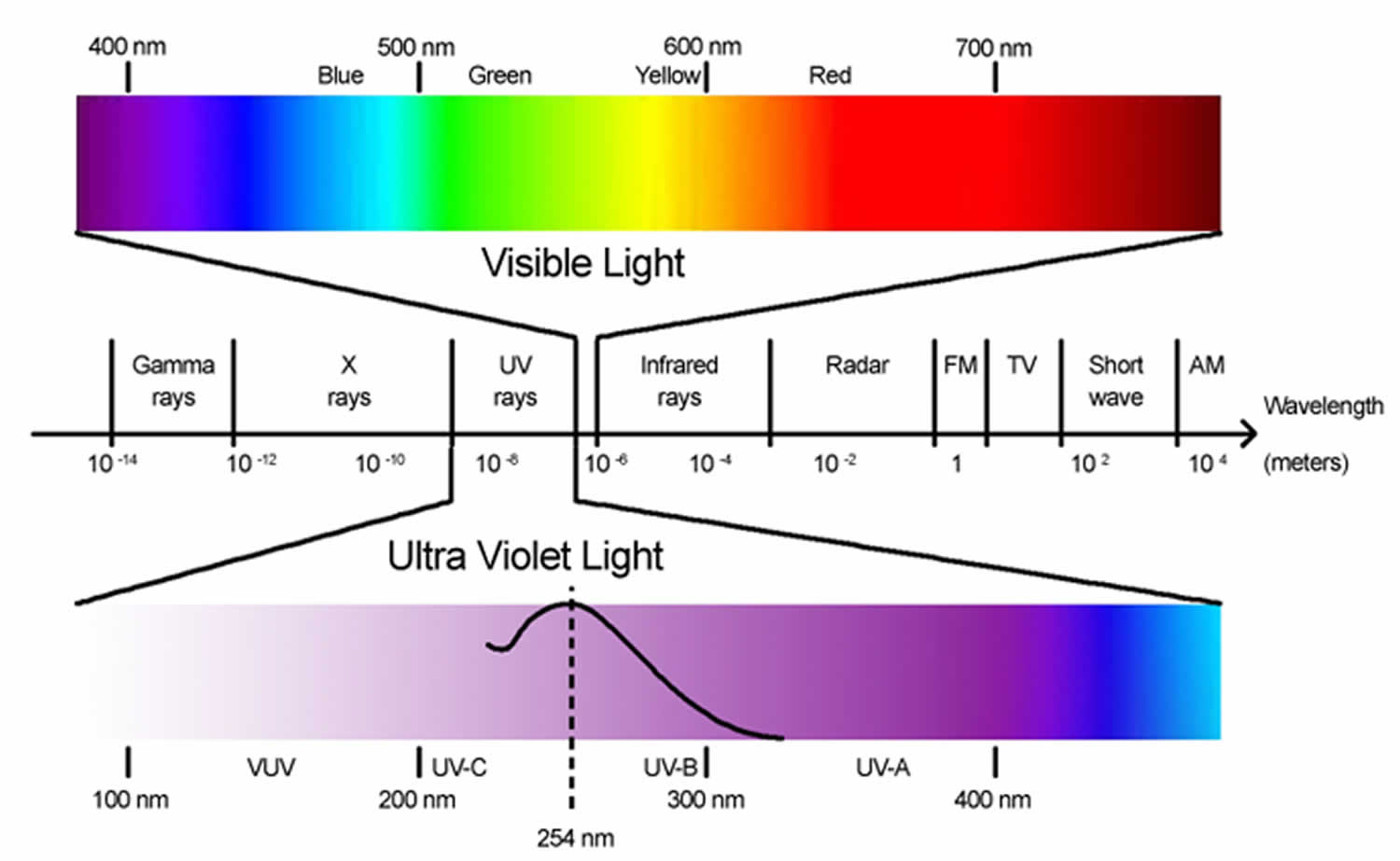
Ultraviolet light is almost as diverse as the various colors of the visible spectrum.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
